|
Sites should be trained in the study specific unbinding procedures at site initiation.A file note should be placed on file to detail the location of the master randomisation schedule until the end of the trial.An audit trail should be available to supporttrack the dissemination and location of the randomisation schedule.
Accidental Unblinding In Clinical Trials Code Break EnvelopesThis may be via code break envelopes with restricted access to dedicated unblinded personnel at sites or via a centralised code break system.
Any tests should be documented in the Investigator Site File (ISF). Uncontrolled clinical observations provide reliable evidence if interventions have dramatic effects along the lines of the effect of insulin in diabetic ketoacidosis. Several quality components for the assessment of bias control have been suggested, but although they seem intrinsically valid, empirical evidence is needed to evaluate their effects on the extent and direction of bias. This narrative review summarizes the findings of methodological studies on the influence of bias in clinical trials. A number of methodological studies suggest that lack of adequate randomization in published trial reports may be associated with more positive estimates of intervention effects. The influence of double-blinding and follow-up is less clear. Several studies have found a significant association between funding sources and pro-industry conclusions. However, the methodological studies also show that bias is difficult to detect and appraise. A-priori exclusion of trials with certain characteristics is not recommended. Appraising bias control in individual trials is necessary to avoid making incorrect conclusions about intervention effects. The potential effect of bias is that investigators will come to the wrong conclusions about the beneficial and harmful effects of interventions. Several mechanisms may bias clinical trials, affecting the estimated intervention effects. Certain trial characteristics have been suggested as quality components that may be used when appraising the control of bias in clinical trials. Although they seem intrinsically valid, empirical evidence is necessary to make conclusions regarding the effects of the individual components. The present paper is a narrative review of methodological studies of bias in clinical trials. Accidental Unblinding In Clinical Trials Manual And ElectronicThe included studies were identified through manual and electronic searches. The manual searches included scanning of bibliographies, journals, and conference proceedings and correspondence with experts. The electronic searches were performed in the Cochrane Library, the Cochrane Hepato-Biliary Group Controlled Trials Register, MEDLINE, and the Excerpta Medica Database (EMBASE). 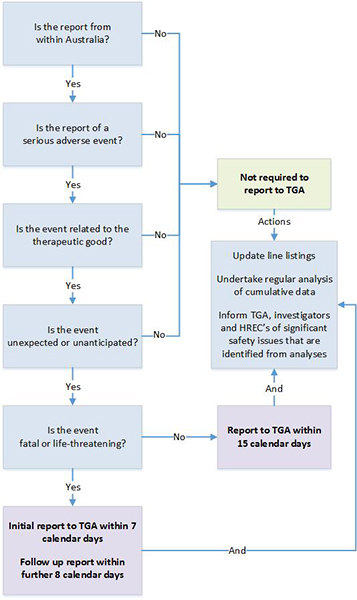
The keywords used in the electronic searches included quality, bias, trials, and random. At least 25 quality scales have been developed, but few have been validated using established criteria ( 3, 4 ). Scales are useful for summarizing the quality of trial cohorts, but the combination of quality components may be problematic. The extent and direction of the effect of individual components may vary, and the conclusions of different scales often disagree ( 4 ). If a significant association between quality and intervention effects is found, separate analyses of the individual components are necessary. Therefore, the use of quality scales is not generally recommended for assessment of bias control in individual trials ( 5 ). The reliability of the evidence depends on several aspects, including the risk of bias associated with different research designs ( 12 ).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- How to speed up utorrent download speed
- Python download file
- Mahabharat star plus all episodes download utorrent
- O download facebook video
- Free itunes download
- Critical ops aimbot apk
- Eca Vrt Disk 2009 Serial
- Mortal kombat google drive download
- Download facebook video downloader
- Clk gtr gtr2 mod
- Fwsim meaning
- Viva caligula in hell game
- Discord download issue
- Portable adobe flash cs6 free download
 RSS Feed
RSS Feed
